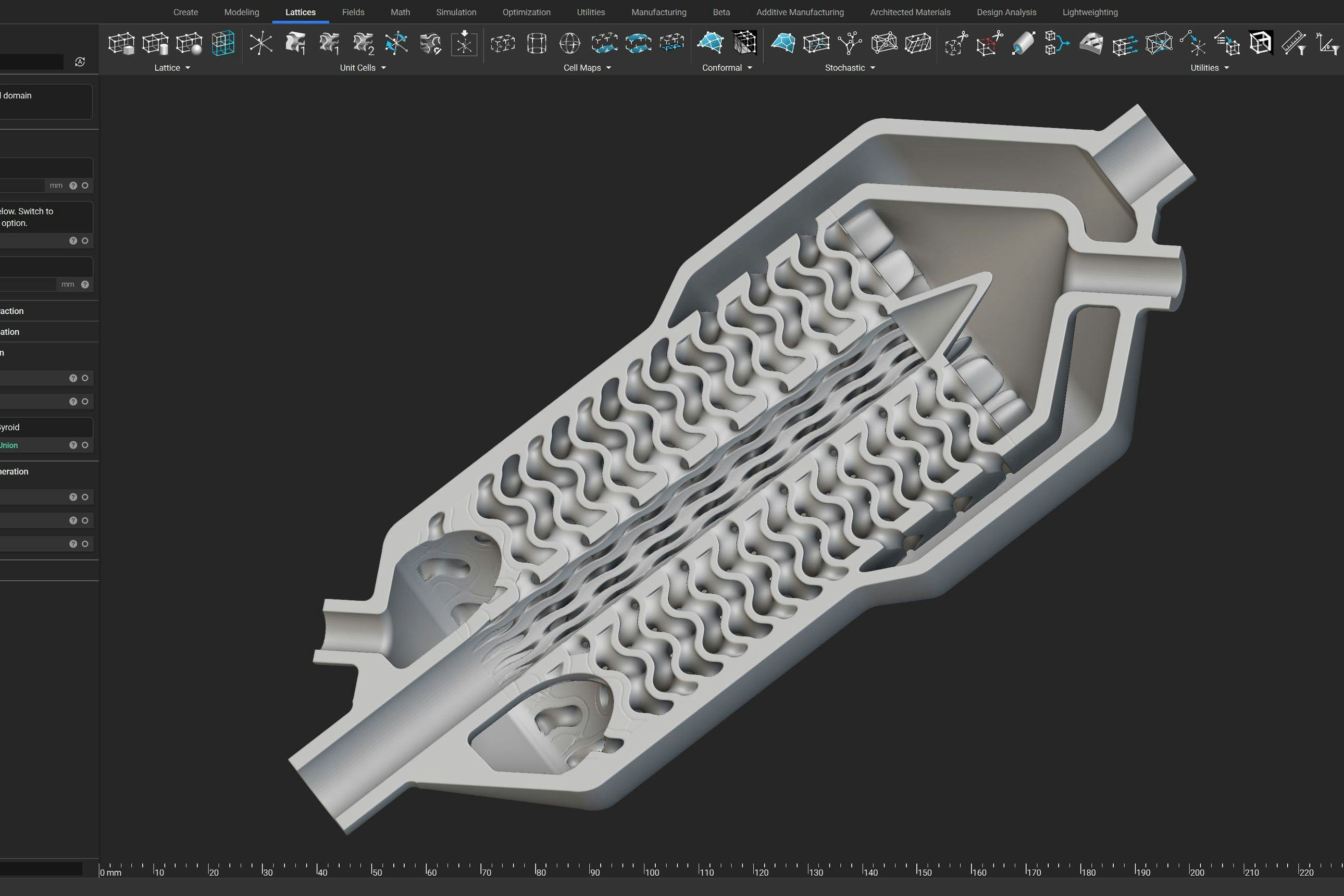
Keeping your medical device design process traceable
Written by nTop
Published on July 9, 2020
When it comes to regulatory oversight, traceability is more a requirement than a recommendation. However, that doesn’t mean implementing it into your processes needs to be an obstacle; nTop offers traceability right out of the box.
A man smarter than myself used to always tell me: “the paperwork is more important than the part”. To this day, I have not encountered a more accurate description of the medical device design process.
A traceable design process means that for any product made, there is a paper trail that keeps track of all the design decisions and input parameters that define that product. Having a traceable process not only improves the credibility of a product, but it ensures that if anything goes wrong, a proper corrective and preventive action can be executed to resolve the issue. Not only is this generally a good practice to follow, but it is also a requirement to ensure your submission survives its review through any of the regulatory bodies. Though this is heavily emphasized during the manufacturing process, the software used is certainly not exempt from these considerations.
nTop takes traceability very seriously and offers a variety of ways to support the medical device industry. When a part is being designed in nTop, the final output that is created is fully defined by the design steps taken to get there. If a single input parameter undergoes a change, the final output is, by definition, different. Though this may appear to be quite obvious, it is not a characteristic that can be fully taken advantage of in standard CAD tools. Though the technical nuances that distinguish traditional BRep modeling and nTop’s implicit modeling can be discussed for days, the biggest takeaway is this: what explicitly defines a design workflow in traditional boundary representation technology can not be as cut-and-dry as it is in nTop's implicit modeling engine. nTop software offers a traceable design process by the sheer nature of how it works.
Reporting
When designing in nTop, the most immediate and noticeable difference in the interface is the Notebook. Where the sidebar would traditionally house a serialized and sequential flow of design operations of which chronology is critical, the Notebook is an environment where multiple design workflows can exist simultaneously. These workflows can originate from a base model and fork into an infinite number of design variations. Or, they can start with separate complex shapes and eventually converge on a single design workflow where one geometry influences the design elements of another. The ability to easily interweave workflows empowers the already heightened design capabilities of topology optimization, finite element analysis, and lattice generation. But at the same time, it also easily enables simple features like embedded reporting.
When a design is being developed for a specific part, a “reporting workflow” can also be implemented simultaneously to capture all the pertinent information that comes into play. As a result, the output of every single design iteration can have an accompanying report generated to ensure that no valuable data is lost in the process.
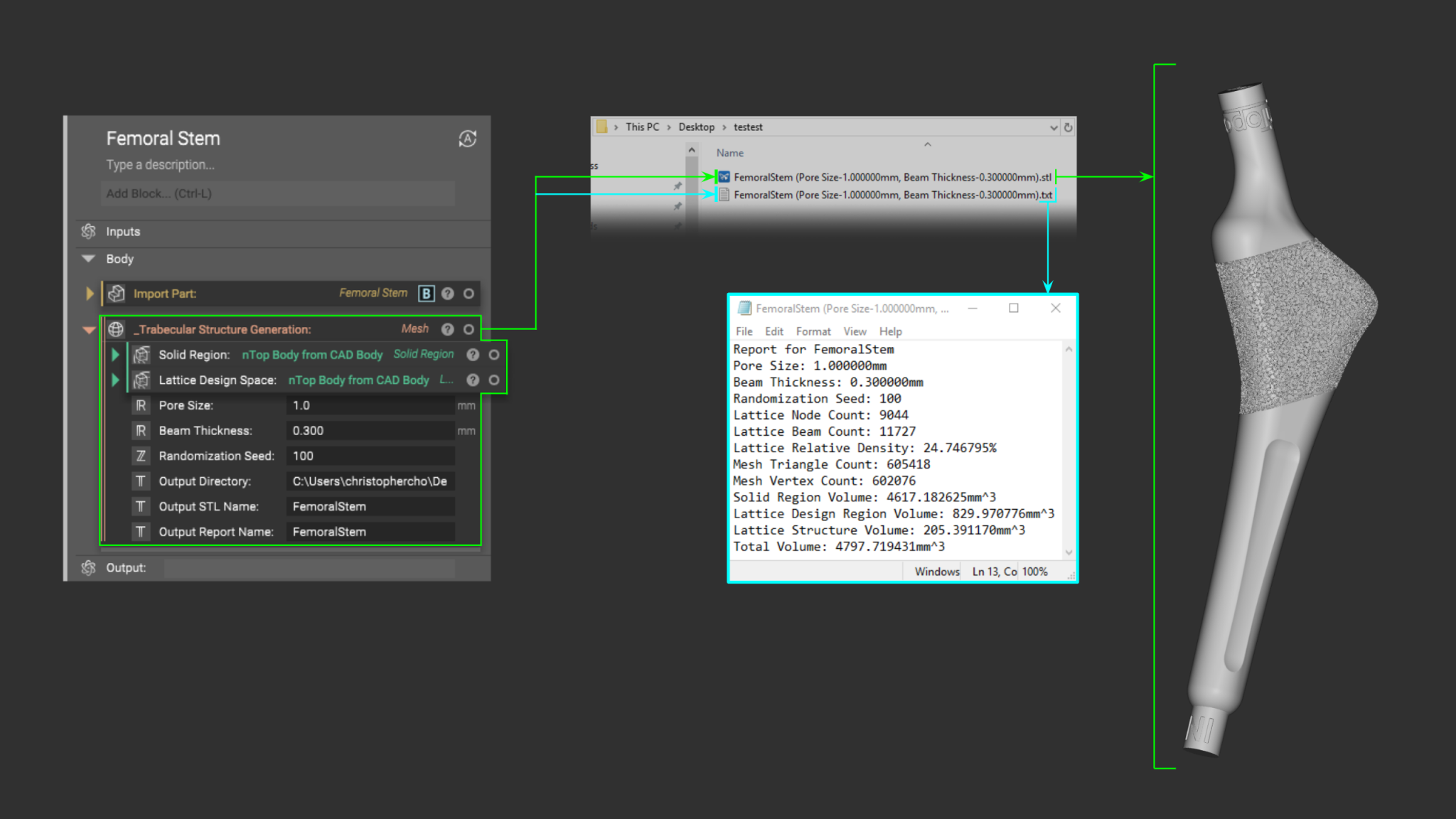
Along with a workflow that uniquely names each exported file, a single notebook becomes a powerful and cohesive collection of workflows that facilitates a traceable process. The value of this ecosystem becomes truly realized when a design process has been finalized and production-ready digital files need to be prepared.
The nTop Notebook becomes an automatable osseointegrative-lattice-generating machine that can churn through an entire part family with accurate file names and traceability reporting in a fraction of the time it would normally take.
Version control
In the regulatory world, change can be frightening. Unprecedented change, however, can be catastrophic. A medical device that has passed initial clinical trials, may still fail inside the human body due to an unexpected change in a critical design area.
A common approach taken to safeguard from this sort of event from occurring is to “lock down” the version of the software tool that was used, ensuring that version is maintained for the development and life of the product. This is a risk management strategy that has been heavily leveraged in the med-device industry to certify that an undocumented or infrastructural change in the way that software works does not impact their product in any damaging way.
If an implant successfully gets through validation, any and all future design revisions must be performed in that version of the software. This seems great from a risk-averse perspective, but it also means that the organization needs to manage and maintain that state of the software in perpetuity. If future versions of that software offer new features or capabilities, that implant is potentially exempt from these benefits.
An approach that nTop has taken to combat this “version freeze” is to offer version control at a more granular level. Because a design workflow is made up of a series of nested “function blocks”, controlling the versions of each of these individual blocks will ensure that your process has stayed exactly the same, even when nTop as a design environment continues to update.
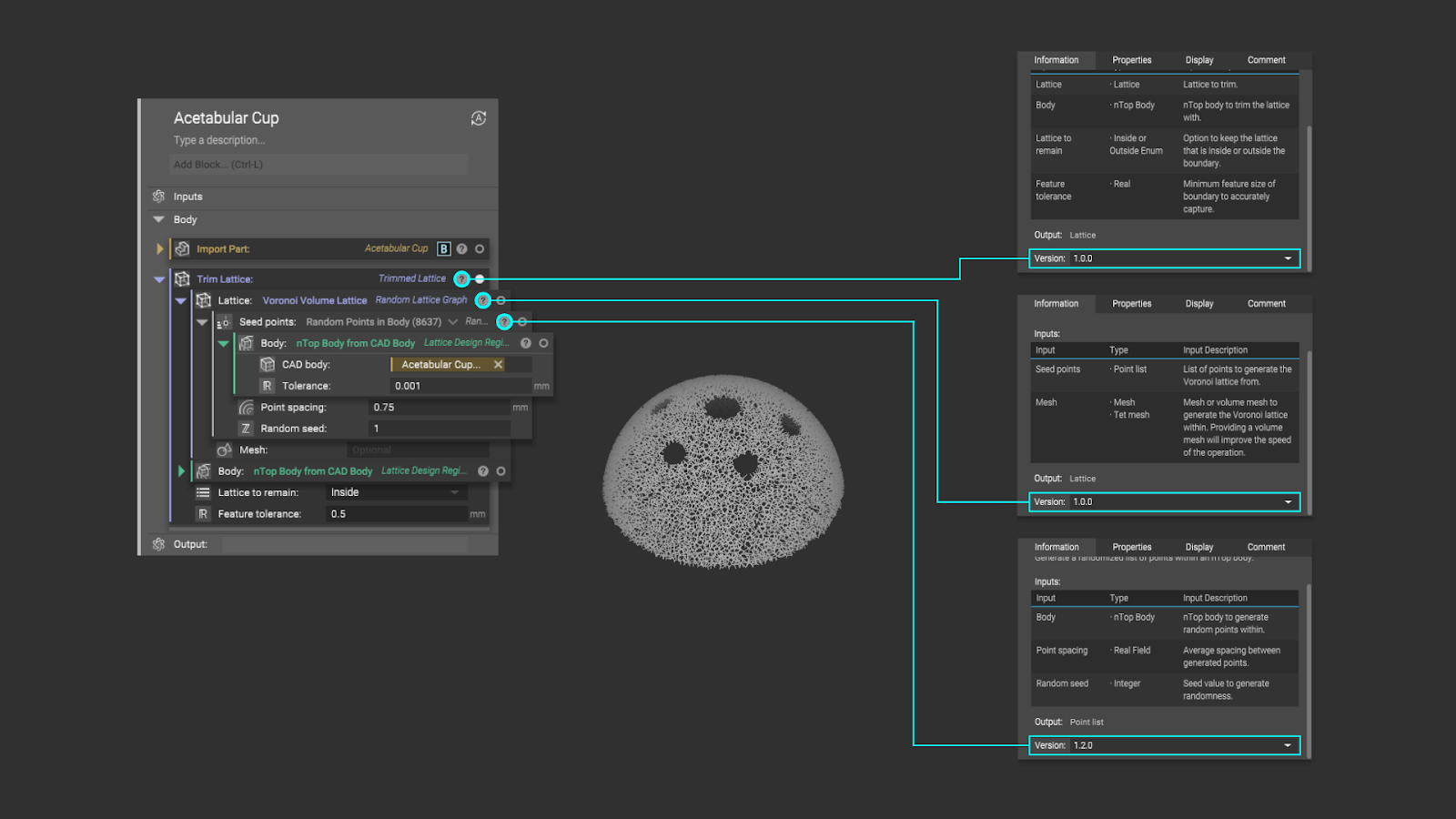
As individual blocks are updated and previous versions become obsolete, the functionality of those old blocks will continue to be available and supported.
However, this doesn’t mean that nTop sees this as a replacement mechanism to the current solution for version control. nTop users still have the freedom to “freeze” their versions. This is just an approach that could make life easier.
If you are looking for additional content on how nTop enables a scalable design approach for orthopedic implants and anatomically-conforming shapes, that allow design workflows to be more accelerated, accurate, and accessible, check out this presentation.

nTop
nTop (formerly nTopology) was founded in 2015 with the belief that engineers’ ability to innovate shouldn’t be limited by their design software. Built on proprietary technologies that upend the constraints of traditional CAD software while integrating seamlessly into existing processes, nTop allows designers in every industry to create complex geometries, optimize instantaneously, and automate workflows to develop breakthrough 3D-printed parts in record time.
Related content
- GUIDE
Download: Advanced design software and additive manufacturing for personalized implants

- VIDEO
Sneak peek into the nTop + Autodesk Fusion 360 integration

- ARTICLE
Optimizing thermal management with conformal cooling to extend operational life
- GUIDE
Download: Design Automation for Medical Devices

- ARTICLE
Advancing structural performance of aerospace heat exchangers